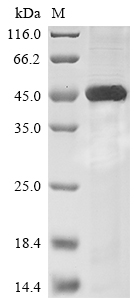
The plasmid vector incorporates the mouse Sialidase-1 (Neu1) protein (42-409aa) encoding gene and the N-terminal 10xHis-tag gene, generating the recombinant plasmid, which is then introduced into E.coli cells. Selection of positive E.coli cells is based on their capacity to withstand a particular antibiotic. Subsequently, the E.coli cells containing the recombinant plasmid are cultivated under conditions that facilitate the expression of the mouse Neu1. Following expression, affinity purification is employed to isolate and purify the recombinant mouse Neu1 protein from the cell lysate. Denaturing SDS-PAGE is used to resolve the resulting recombinant protein, enabling an estimation of its purity, exceeding 85%.
Neu1, also known as neuraminidase 1, is an enzyme found in lysosomes, essential for breaking down sialylated glycoconjugates [1]. It's particularly effective against short sugar chains and small protein-sugar complexes [1]. Neu1 shows increased activity when monocytes transform into macrophages, indicating its importance in macrophage function and immune response [2]. It's also involved in regulating insulin signaling [3], managing energy use and stress in heart cells after a heart attack [4], and promoting insulin signaling [5]. Moreover, Neu1 seems to influence immune response, tissue elasticity, and cell growth [5]. It can even alter the signals of Toll-like receptors, impacting the response to certain infections [6]. In some cancers, like melanoma, Neu1 may have a role, although it's not fully clear yet [7]. This enzyme is made in the endoplasmic reticulum, then modified and activated with the help of another protein, protective protein/cathepsin A (PPCA) [8][9][10]. Neu1 also affects microglial activation and contributes to inflammatory processes in diseases like aortic dissection [11][12]. In certain bladder cancers, Neu1 slows down cancer cell growth by interfering with specific protein interactions and signaling pathways [13]. It's also linked to changes in receptors that control the release of fat-related hormones, hinting at a role in regulating fat tissue functions [14].
References:
[1] V. Seyrantepe, K. Landry, S. Trudel, J. Hassan, C. Morales, & A. Pshezhetsky, "Neu4, a novel human lysosomal lumen sialidase, confers normal phenotype to sialidosis and galactosialidosis cells", Journal of Biological Chemistry, vol. 279, no. 35, p. 37021-37029, 2004. https://doi.org/10.1074/jbc.m404531200
[2] F. Liang, V. Seyrantepe, K. Landry, R. Ahmad, A. Ahmad, N. Stamatoset al., "Monocyte differentiation up-regulates the expression of the lysosomal sialidase, neu1, and triggers its targeting to the plasma membrane via major histocompatibility complex class ii-positive compartments", Journal of Biological Chemistry, vol. 281, no. 37, p. 27526-27538, 2006. https://doi.org/10.1074/jbc.m605633200
[3] L. Dridi, V. Seyrantepe, A. Fougerat, X. Pan, É. Bonneil, P. Thibaultet al., "Positive regulation of insulin signaling by neuraminidase 1", Diabetes, vol. 62, no. 7, p. 2338-2346, 2013. https://doi.org/10.2337/db12-1825
[4] Z. Guo, D. Fan, F. Liu, S. Ma, P. An, D. Yanget al., "Neu1 regulates mitochondrial energy metabolism and oxidative stress post-myocardial infarction in mice via the sirt1/pgc-1 alpha axis", Frontiers in Cardiovascular Medicine, vol. 9, 2022. https://doi.org/10.3389/fcvm.2022.821317
[5] A. Khan and C. Sergi, "Neu1—a unique therapeutic target for alzheimer’s disease", Frontiers in Pharmacology, vol. 13, 2022. https://doi.org/10.3389/fphar.2022.902259
[6] J. Karmakar, S. Roy, & C. Mandal, "Modulation of tlr4 sialylation mediated by a sialidase neu1 and impairment of its signaling in leishmania donovani infected macrophages", Frontiers in Immunology, vol. 10, 2019. https://doi.org/10.3389/fimmu.2019.02360
[7] P. Qiu, L. Gao, H. Cheng, J. Wang, & J. Wang, "Sialidase neu1 may serve as a potential biomarker of proliferation, migration and prognosis in melanoma", World Journal of Oncology, vol. 13, no. 4, p. 222-234, 2022. https://doi.org/10.14740/wjon1509
[8] J. Tsukimoto, Y. Takeuchi, Y. Horii, T. Iniwa, Y. Fukushi, S. Nishiokaet al., "In cellulo crystallization of human neuraminidase 1 and biological roles of n-glycans", Acs Applied Bio Materials, vol. 5, no. 1, p. 205-213, 2021. https://doi.org/10.1021/acsabm.1c01043
[9] E. Bonten, G. Yogalingam, H. Hu, E. Gomero, D. Vlekkert, & A. d’Azzo, "Chaperone-mediated gene therapy with recombinant aav-ppca in a new mouse model of type i sialidosis", Biochimica Et Biophysica Acta (Bba) - Molecular Basis of Disease, vol. 1832, no. 10, p. 1784-1792, 2013. https://doi.org/10.1016/j.bbadis.2013.06.002
[10] D. Wang, S. Zaitsev, G. Taylor, A. d'Azzo, & E. Bonten, "Protective protein/cathepsin a rescues n-glycosylation defects in neuraminidase-1", Biochimica Et Biophysica Acta (Bba) - General Subjects, vol. 1790, no. 4, p. 275-282, 2009. https://doi.org/10.1016/j.bbagen.2009.01.006
[11] D. Allendorf, E. Franssen, & G. Brown, "Lipopolysaccharide activates microglia via neuraminidase 1 desialylation of toll‐like receptor 4", Journal of Neurochemistry, vol. 155, no. 4, p. 403-416, 2020. https://doi.org/10.1111/jnc.15024
[12] Q. Wang, Z. Chen, X. Peng, Z. Zheng, A. Le, J. Guoet al., "Neuraminidase 1 exacerbating aortic dissection by governing a pro-inflammatory program in macrophages", Frontiers in Cardiovascular Medicine, vol. 8, 2021. https://doi.org/10.3389/fcvm.2021.788645
[13] X. Zhou, Y. Zhai, C. Liu, G. Yang, J. Guo, G. Liet al., "Sialidase neu1 suppresses progression of human bladder cancer cells by inhibiting fibronectin-integrin α5β1 interaction and akt signaling pathway", Cell Communication and Signaling, vol. 18, no. 1, 2020. https://doi.org/10.1186/s12964-019-0500-x
[14] Y. Natori, N. Ohkura, M. Nasui, G. Atsumi, & F. Kihara-Negishi, "Acidic sialidase activity is aberrant in obese and diabetic mice", Biological and Pharmaceutical Bulletin, vol. 36, no. 6, p. 1027-1031, 2013. https://doi.org/10.1248/bpb.b12-00995